|
The formula for determining the hybridization of any substance is: There is an image linked below for your convenience. Specifically, one electron in the 3p orbital and another electron in the 3d orbital established the bonds between sulphur and oxygen. And the remaining two pairs of orbitals make up sulfur’s lone pair.Īre you curious about the two 3p electrons that were not involved in hybridization? Sulfur requires two unpaired electrons from the Sp2 hybridised orbitals in order to create two sigma bonds with oxygen. Now, 3s2 and 3p3 combine to generate Sp2 hybridization, which consists of 3 analogous orbitals with 2 paired and 2 unpaired electrons. When an electron is in an excited condition, it jumps from 3px to 3d orbital. 
Similarly, the ground state electronic arrangement of SO2 is 1s2 2s2 2p6 3s2 3s4. I would recommend understanding the theory first, then proceeding to the formal.Ī brief tip: when one s orbital combines with two p orbitals, Sp2 hybridization results in three equal s orbitals. Now, hybridization of SO2 can be comprehended in two ways: the first is through theory, and the second is through formula application. Next, we must learn about the hybridization of SO2. Lastly, remember to check the formal charge of each atom! Oxygen has two lone pairs, while sulphur has one. This will eventually complete the atom’s octet. 
We must position these remaining electrons around the atoms in accordance with the specifications. Subtracting that from the total number of valence electrons, we are left with 10 electrons. Consequently, the amount of electrons utilised in double bonds = 8 The molecule contains 2 oxygen atoms, therefore = 6*2 = 12Īfter drawing the skeletal structure, it is evident that no atoms can complete their octet with single bonds. In SO2, the valence electron of sulphur equals six. Using the following methods, formal charge calculations can be performed: Step 6 – Finally, it is essential to ensure that all atoms have the lowest feasible formal charge. Step 5: If double or triple bonds are required to satisfy the octet rule for all atoms, they must be formed. Always start with the electronegative atoms and then proceed to the electropositive atoms. The third stage is to create a skeleton structure with only single bonds.Īfter forming single bonds, the fourth step involves completing the octet of the atoms with the leftover electrons. The central atom is the atom with the maximum number of bonding sites. The second step is to identify the core atom. A ‘+’ signifies electron loss, whereas a ‘-‘ signifies electron gain. While doing so, be mindful of the + and – symbols. Step 1 – The first and most crucial step is determining the total amount of valence electrons in the molecule. Now let’s examine the process of drawing lewis structure: This structure allows us to determine the type and amount of bonds that comprise the molecule. The distribution of electrons around the atoms of a chemical is its Lewis structure. Now we will discuss essential concepts such as Lewis structure, molecular geometry, the MO Diagram, and SO2 hybridization.īefore diving into the lewis structure of SO2, let’s briefly explore the significance of lewis structure and the methods required to draw it. 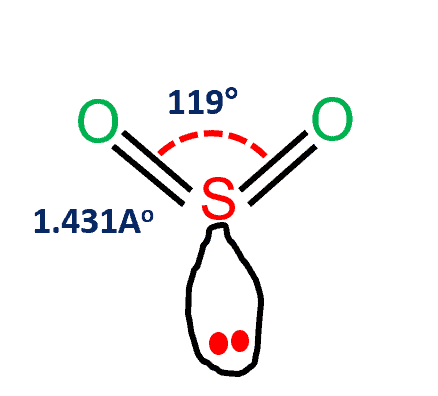
The melting point and boiling point are, respectively, -72☌ and -10☌. In terms of its characteristics, the molar mass of SO2 is 64,066 g/mol. In Commonwealth English, sulphur dioxide is spelt as sulphur dioxide. Because at the end, you will have a thorough understanding of the fundamentals of SO2 before moving on to the reactions. However, before reading through them, please read the following article. In addition to its primary function, which is the generation of sulfuric acid, SO2 has various purposes in the chemical industry. Have you ever wondered what generates the scent of burning matchsticks? The correct response is SO2!
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
